 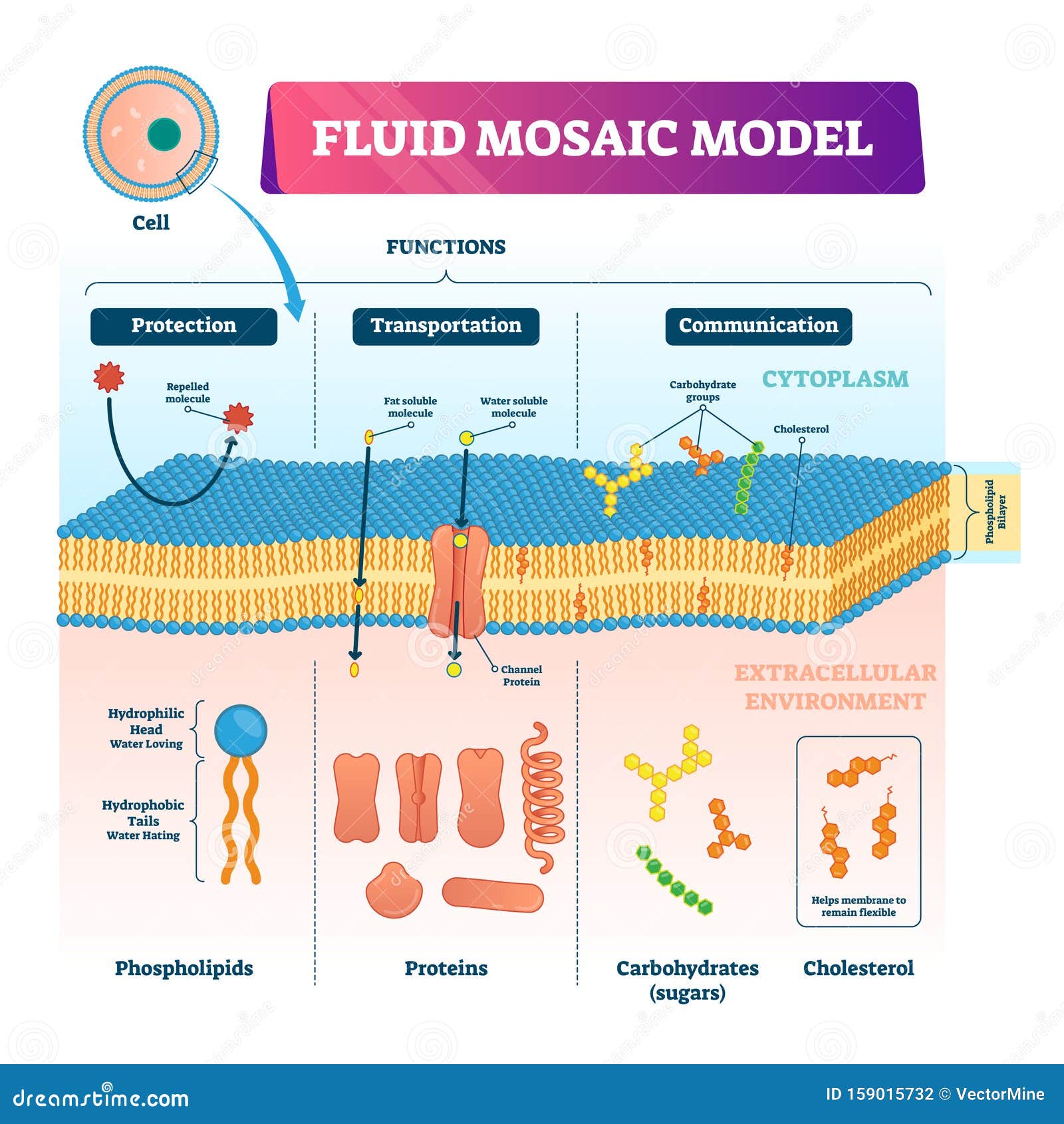
CholesterolĬholesterol is vital for many functions in a cell, including very importantly, a major constituent of the cell membrane.Ĭholesterol itself consists of a polar head, a planar steroid ring and a non-polar hydrocarbon tail. In the bilayer, there are van der Waal forces between the fatty acid tails of the phospholipid, with electrostatic and hydrogen bonds between the hydrophilic groups and water.įig 2 – Diagram showing the structure of both the phospholipid bilayer and an individual phospholipid. They spontaneously form bilayers in the water with the head groups facing out and the tail groups facing in. 
Phospholipid molecules are therefore amphipathic – being both hydrophilic and hydrophobic. As the tail is made of fatty acids, it does not form hydrogen bonds with water and therefore is hydrophobic and non-polar. If the chain contains a cis double bond then the chain is kinked – therefore reducing the tight packing of the membrane and so increasing its movement. Tail of 2 fatty acid chains – normally consisting of between 14-24 carbons (but the most common carbon lengths are 16 and 18).a sugar or choline – meaning that the head end of the phospholipid is hydrophilic. Head group- This is a polar group e.g. /cell-membrane--artwork-547999983-5a942e7a875db900363afbf1.jpg)
These consist of a head molecule, a phosphate molecule, a glycerol and two fatty acid chains. The membrane bilayer contains many kinds of phospholipid molecules, with different sized head and tail molecules. – Often found attached to proteins/lipids on the outside of the cell membrane – a coat of carbohydrate surrounding a cell is often called the glycocalyx A simplified rough guide of the dry weight is shown in Table 1. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
